- Volumes 108-119 (2025)
-
Volumes 96-107 (2025)
-
Volume 107
Pages 1-376 (December 2025)
-
Volume 106
Pages 1-336 (November 2025)
-
Volume 105
Pages 1-356 (October 2025)
-
Volume 104
Pages 1-332 (September 2025)
-
Volume 103
Pages 1-314 (August 2025)
-
Volume 102
Pages 1-276 (July 2025)
-
Volume 101
Pages 1-166 (June 2025)
-
Volume 100
Pages 1-256 (May 2025)
-
Volume 99
Pages 1-242 (April 2025)
-
Volume 98
Pages 1-288 (March 2025)
-
Volume 97
Pages 1-256 (February 2025)
-
Volume 96
Pages 1-340 (January 2025)
-
Volume 107
-
Volumes 84-95 (2024)
-
Volume 95
Pages 1-392 (December 2024)
-
Volume 94
Pages 1-400 (November 2024)
-
Volume 93
Pages 1-376 (October 2024)
-
Volume 92
Pages 1-316 (September 2024)
-
Volume 91
Pages 1-378 (August 2024)
-
Volume 90
Pages 1-580 (July 2024)
-
Volume 89
Pages 1-278 (June 2024)
-
Volume 88
Pages 1-350 (May 2024)
-
Volume 87
Pages 1-338 (April 2024)
-
Volume 86
Pages 1-312 (March 2024)
-
Volume 85
Pages 1-334 (February 2024)
-
Volume 84
Pages 1-308 (January 2024)
-
Volume 95
-
Volumes 72-83 (2023)
-
Volume 83
Pages 1-258 (December 2023)
-
Volume 82
Pages 1-204 (November 2023)
-
Volume 81
Pages 1-188 (October 2023)
-
Volume 80
Pages 1-202 (September 2023)
-
Volume 79
Pages 1-172 (August 2023)
-
Volume 78
Pages 1-146 (July 2023)
-
Volume 77
Pages 1-152 (June 2023)
-
Volume 76
Pages 1-176 (May 2023)
-
Volume 75
Pages 1-228 (April 2023)
-
Volume 74
Pages 1-200 (March 2023)
-
Volume 73
Pages 1-138 (February 2023)
-
Volume 72
Pages 1-144 (January 2023)
-
Volume 83
-
Volumes 60-71 (2022)
-
Volume 71
Pages 1-108 (December 2022)
-
Volume 70
Pages 1-106 (November 2022)
-
Volume 69
Pages 1-122 (October 2022)
-
Volume 68
Pages 1-124 (September 2022)
-
Volume 67
Pages 1-102 (August 2022)
-
Volume 66
Pages 1-112 (July 2022)
-
Volume 65
Pages 1-138 (June 2022)
-
Volume 64
Pages 1-186 (May 2022)
-
Volume 63
Pages 1-124 (April 2022)
-
Volume 62
Pages 1-104 (March 2022)
-
Volume 61
Pages 1-120 (February 2022)
-
Volume 60
Pages 1-124 (January 2022)
-
Volume 71
- Volumes 54-59 (2021)
- Volumes 48-53 (2020)
- Volumes 42-47 (2019)
- Volumes 36-41 (2018)
- Volumes 30-35 (2017)
- Volumes 24-29 (2016)
- Volumes 18-23 (2015)
- Volumes 12-17 (2014)
- Volume 11 (2013)
- Volume 10 (2012)
- Volume 9 (2011)
- Volume 8 (2010)
- Volume 7 (2009)
- Volume 6 (2008)
- Volume 5 (2007)
- Volume 4 (2006)
- Volume 3 (2005)
- Volume 2 (2004)
- Volume 1 (2003)
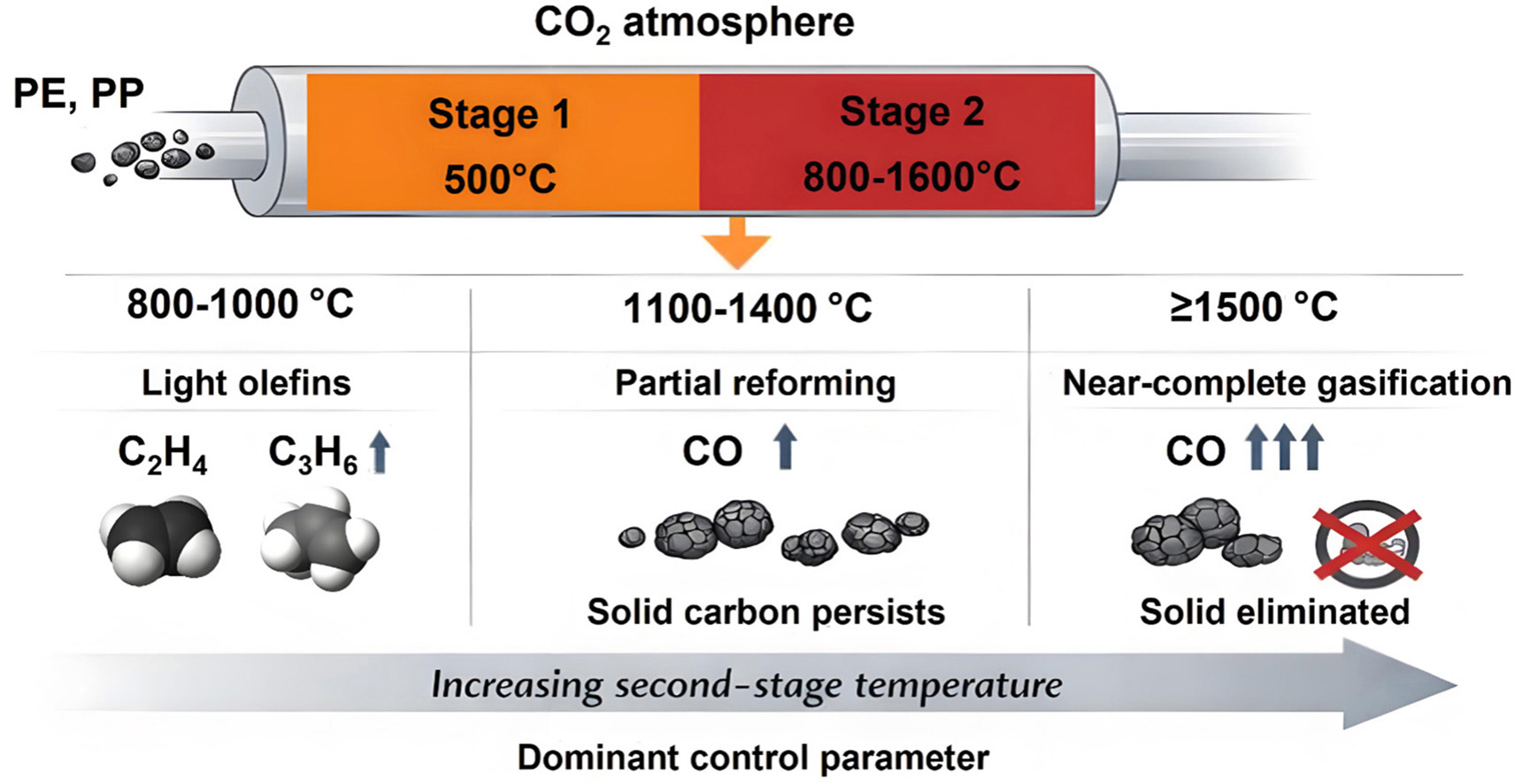
• Temperature governs gaseous product distribution during polyolefin pyrolysis.
• At moderate temperatures the introduction of CO2 reduces light olefin yields by promoting secondary reactions.
• Near-complete conversion into CO is realized when the second-stage temperature exceeds 1500 °C.
Chemical recycling of polyolefin plastics is a key approach to mitigating plastic pollution and enabling carbon resource circularity. In this work, the pyrolysis behaviors and product distributions of polyethylene (PE) and polypropylene (PP) are systematically investigated under N2 and CO2 atmospheres over a wide temperature range of 500–1600 °C using thermogravimetric analysis (TGA) and a two-stage tubular reactor. TGA results indicate that below 500 °C, CO2 behaves similarly to N2 as a physically inert carrier gas and does not affect the primary thermal decomposition of polyolefins. Two-stage pyrolysis experiments reveal the temperature thresholds for the chemical activation of CO2, at which CO2 starts to participate in reactions with pyrolysis intermediates, occurring at approximately 900 °C for PP and 1100 °C for PE, as evidenced by the formation of CO. Within the temperature range of 800–1000 °C, light olefins such as ethylene and propylene dominate the gaseous products; however, the introduction of CO2 leads to a slight decrease in light olefin yields compared with those obtained under N2. At higher temperatures of 1100–1400 °C, light olefins undergo extensive secondary cracking and reforming reactions, resulting in a pronounced shift of products toward syngas, accompanied by significant solid carbon deposition. When the temperature is further increased above 1500 °C, carbon deposition is effectively eliminated, and near-complete conversion of PE and PP into syngas, predominantly CO, is achieved. These results demonstrate the feasibility of switching product selectivity from light olefin recovery to syngas production within a single reaction system through temperature regulation, providing important insights for the design of high-value chemical recycling processes for waste polyolefin plastics.